The 4-State model of sarcomeric contraction created by Landesberg and Sideman is investigated in terms of the energy liberated by the system as a function of the rate of shortening.
Description
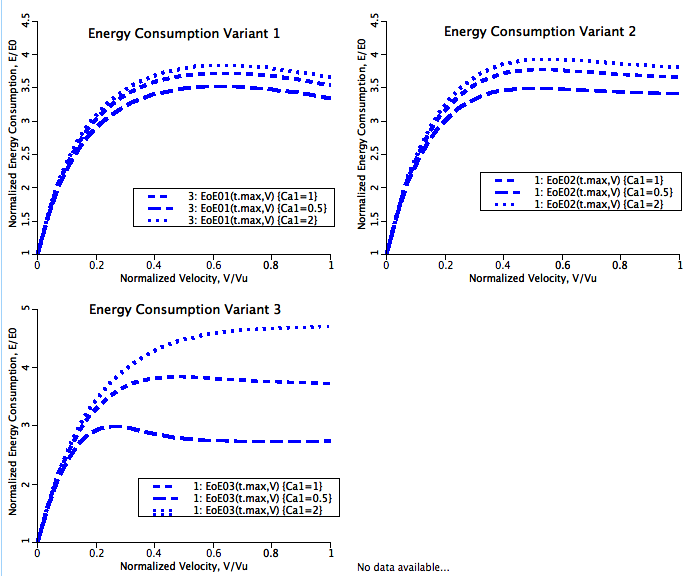
This model characterizes the energy liberation rate during steady sarcomeric contraction as developed by Tchaicheeyan and Landesberg (Am J Physiol 289:H2176-H2182, 2005). Three hypotheses are applied to the basic Landesberg 4-State model of sarcomeric function in an effort to explain the biphasic energy liberation rate - velocity relationship. They are:
1) Crossbridge turnover rate from non-force generating (weak) to force generating (strong) conformation decreases as velocity increases.
2) Crossbridge kinetics is determined by the number of strong crossbridges
3) The affinity of troponin for calcium is modulated by the number of strong crossbridges.
All three hypotheses yeild biphasic energy liberation rate - velocity relationships. The equations given below show how these three hypotheses are integrated into the basic Landesberg 4-State model.
Equations
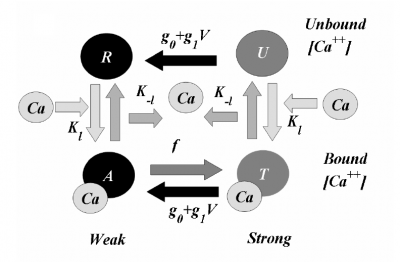
The basic 4-State sarcomere model relates force generation to the two strong states of troponin. The set of ODEs governing the transition of the troponin regulatory unit through its four states is given by:
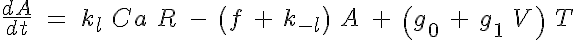
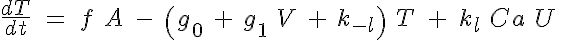
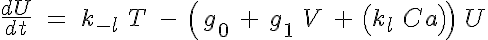
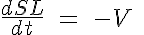
and
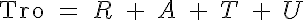
where R, A, T and U are the unbound weak, bound weak, bound strong and unbound strong states of troponin, respectively. Tro is the total concentration of troponin regulatory units, SL is the sarcomere length and V is the sarcomere contraction velocity defined as positive in the case of sarcomere shortening. The three variants of the model affect the kinetics of the basic 4-State model in the following ways: Variant 1: Velocity affecting weak to strong transition rate
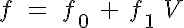
Variant 2: Crossbridge - crossbridge cooperativity
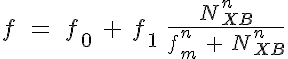
Variant 3: Crossbridge - calcium cooperativity
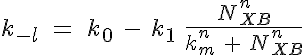
where f is the weak to strong transition rate of troponin, k-l is the calcium bound to unbound transition rate, NXB is the number of strong crossbridges, and fm and km represent the number of strong crossbridges which represents an f or k-1, respectively, at the midpoint of its range.
- Download JSim model MML code (text):
- Download translated SBML version of model (if available):
- No SBML translation currently available.
- Information on SBML conversion in JSim
We welcome comments and feedback for this model. Please use the button below to send comments:
Tchaicheeyan O and Landesberg A.
Regulation of energy liberation during steady sarcomere shortening.
American Journal of Physiology, Heart and Circulatory Physiology 289:H2176-H2182, 2005.
Landesberg A and Sideman S.
Force-velocity relationship and biochemical-to-mechanical energy conversion by the sarcomere.
American Journal of Physiology, Heart and Circulatory Physiology 278:H1274-H1284, 2000.
Please cite https://www.imagwiki.nibib.nih.gov/physiome in any publication for which this software is used and send one reprint to the address given below:
The National Simulation Resource, Director J. B. Bassingthwaighte, Department of Bioengineering, University of Washington, Seattle WA 98195-5061.
Model development and archiving support at https://www.imagwiki.nibib.nih.gov/physiome provided by the following grants: NIH U01HL122199 Analyzing the Cardiac Power Grid, 09/15/2015 - 05/31/2020, NIH/NIBIB BE08407 Software Integration, JSim and SBW 6/1/09-5/31/13; NIH/NHLBI T15 HL88516-01 Modeling for Heart, Lung and Blood: From Cell to Organ, 4/1/07-3/31/11; NSF BES-0506477 Adaptive Multi-Scale Model Simulation, 8/15/05-7/31/08; NIH/NHLBI R01 HL073598 Core 3: 3D Imaging and Computer Modeling of the Respiratory Tract, 9/1/04-8/31/09; as well as prior support from NIH/NCRR P41 RR01243 Simulation Resource in Circulatory Mass Transport and Exchange, 12/1/1980-11/30/01 and NIH/NIBIB R01 EB001973 JSim: A Simulation Analysis Platform, 3/1/02-2/28/07.

