Physiological model of the full baroreflex heart control system based on experimental measurements
Description
This is a phenomenological ODE model of baroreflex open-loop control of heart rate. An aortic blood pressure signal (the driving signal for the model) is transduced by afferent baroreceptor nerve fibers in the wall of the aortic arch into a neural (firing rate) signal. This transduction process is governed by mechanical strain in the wall of the aorta. This neural signal is then relayed and further processed by the central and peripheral nervous systems into parallel sympathetic and parasympathetic tone signals. Sympathetic and parasympathetic tones drive release of norepinephrine and acetylcholine, respectively, into the neuromuscular junction of the sinoatrial node of the heart, thereby modulating the pacemaker activity at the sinoatrial node. The sinoatrial node is the ultimate effector of heart rate, which is the model output. Parameters used to reproduce the figures in the paper were taken from Table 5 of the paper. All pressure data was taken over a period of 120 seconds at a rate of 100 samples per second. Heart rate data was derived from the pressure data using a high-pass filter while detecting the upstroke zero-crossings. LIMITATIONS The model cannot fully account for the fast dynamics of the parasympathetic nervous system. Additionally, time delays in the peripheral nervous system are assumed to be negligible and have been omitted. ERRATA The caption for Figure 8 in the paper erroneously states: "Data shown in A and B are from the SS and SS.13BN strains, respectively." and should read: "Data shown in A and B are from the SS.13BN and SS strains, respectively." The strain attributions were transposed; 8A is from SS.13BN along with its derived graphs, C and E, while 8B is from Dahl SS along with D and F. The plots contained in this project are labeled correctly.
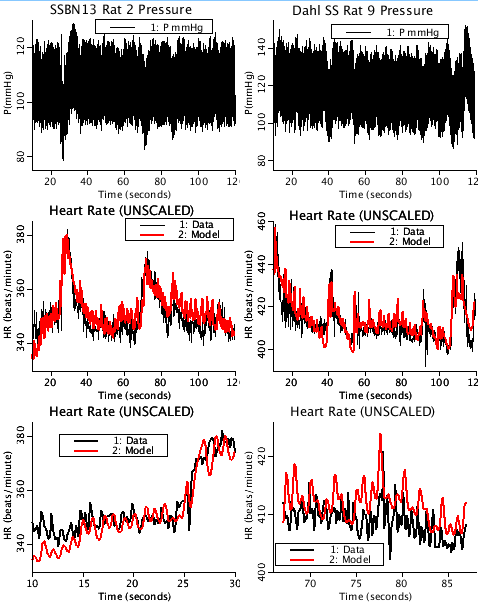
This figure is similar to Figure 8 in the referenced publication.
Equations
None.
The equations for this model may be viewed by running the JSim model applet and clicking on the Source tab at the bottom left of JSim's Run Time graphical user interface. The equations are written in JSim's Mathematical Modeling Language (MML). See the Introduction to MML and the MML Reference Manual. Additional documentation for MML can be found by using the search option at the Physiome home page.
- Download JSim model MML code (text):
- Download translated SBML version of model (if available):
We welcome comments and feedback for this model. Please use the button below to send comments:
Scott M. Bugenhagen, Allen W. Cowley, Jr. and Daniel A. Beard. Identifying physiological origins of baroreflex dysfunction in salt-sensitive hypertension in the Dahl SS rat.Physiol. Genomics 42:23-41, 2010.
Please cite https://www.imagwiki.nibib.nih.gov/physiome in any publication for which this software is used and send one reprint to the address given below:
The National Simulation Resource, Director J. B. Bassingthwaighte, Department of Bioengineering, University of Washington, Seattle WA 98195-5061.
Model development and archiving support at https://www.imagwiki.nibib.nih.gov/physiome provided by the following grants: NIH U01HL122199 Analyzing the Cardiac Power Grid, 09/15/2015 - 05/31/2020, NIH/NIBIB BE08407 Software Integration, JSim and SBW 6/1/09-5/31/13; NIH/NHLBI T15 HL88516-01 Modeling for Heart, Lung and Blood: From Cell to Organ, 4/1/07-3/31/11; NSF BES-0506477 Adaptive Multi-Scale Model Simulation, 8/15/05-7/31/08; NIH/NHLBI R01 HL073598 Core 3: 3D Imaging and Computer Modeling of the Respiratory Tract, 9/1/04-8/31/09; as well as prior support from NIH/NCRR P41 RR01243 Simulation Resource in Circulatory Mass Transport and Exchange, 12/1/1980-11/30/01 and NIH/NIBIB R01 EB001973 JSim: A Simulation Analysis Platform, 3/1/02-2/28/07.

