Rate Dependence and Regulation of Action Potential and Calcium Transient in a Ventricular Cell Model. Based on the Hund-Rudy 2004 model. Imported from the CellML version of the model.
Description
In the paper described here, Thomas Hund and Yoram Rudy present a detailed, and physiologically realistic, mathematical model of a canine ventricular cell. Model simulations are able to recreate the rate-dependent phenomena associated with ion-channel kinetics, action potential properties, and calcium ion handling. The model is based on an epicardial myocyte because these cells contain the largest transient outward potassium current (when compared with endocardial or midmyocardial myocytes). The calcium/calmodulin-dependent protein kinase (CaMKII) regulatory pathway was embedded within the electrophysiological model, incorporating calcium-release formulation, calcium subspace, and dynamic chloride handling. Results from the model simulations revealed CaMKII is an important determinant of the rate dependence of the calcium transient, but not of the action potential duration, which depends instead on the ion-channel kinetics. * Original CellML note: * This CellML version of the model has been checked in COR and * OpenCell. The units are consistent and the model runs to recreate * the published results. This is the tissue version of the model, * to use this model embedded within tissue, set the initial * value of the variable tissue in component Environment to 1.
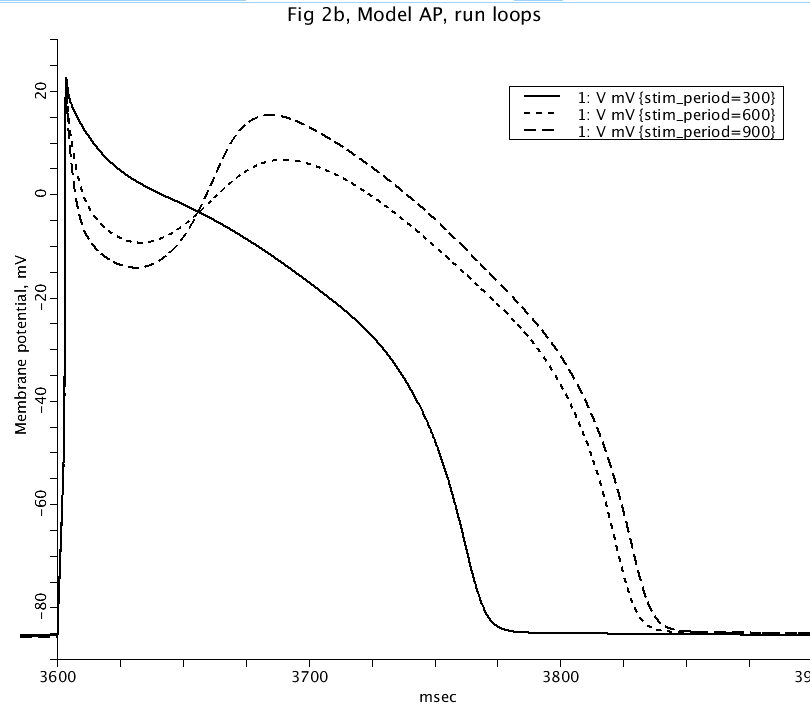
Figure: Action potential at steady-state for different action potential cycle lengths of 300, 600, and 900 ms.
Equations
The equations for this model may be viewed by running the JSim model applet and clicking on the Source tab at the bottom left of JSim's Run Time graphical user interface. The equations are written in JSim's Mathematical Modeling Language (MML). See the Introduction to MML and the MML Reference Manual. Additional documentation for MML can be found by using the search option at the Physiome home page.
- Download JSim model MML code (text):
- Download translated SBML version of model (if available):
We welcome comments and feedback for this model. Please use the button below to send comments:
Thomas J. Hund and Yoram Rudy, Rate dependence and regulation of action potential and calcium transient in a canine cardiac ventricular cell model, 2004,Circ,110, 3168-3174. PubMedID: 15505083 Luo CH, Rudy Y. A dynamic model of the cardiac ventricular action potential, I: simulations of ionic currents and concentration changes. Circ Res. 1994;74:1071–1096.
Please cite https://www.imagwiki.nibib.nih.gov/physiome in any publication for which this software is used and send one reprint to the address given below:
The National Simulation Resource, Director J. B. Bassingthwaighte, Department of Bioengineering, University of Washington, Seattle WA 98195-5061.
Model development and archiving support at https://www.imagwiki.nibib.nih.gov/physiome provided by the following grants: NIH U01HL122199 Analyzing the Cardiac Power Grid, 09/15/2015 - 05/31/2020, NIH/NIBIB BE08407 Software Integration, JSim and SBW 6/1/09-5/31/13; NIH/NHLBI T15 HL88516-01 Modeling for Heart, Lung and Blood: From Cell to Organ, 4/1/07-3/31/11; NSF BES-0506477 Adaptive Multi-Scale Model Simulation, 8/15/05-7/31/08; NIH/NHLBI R01 HL073598 Core 3: 3D Imaging and Computer Modeling of the Respiratory Tract, 9/1/04-8/31/09; as well as prior support from NIH/NCRR P41 RR01243 Simulation Resource in Circulatory Mass Transport and Exchange, 12/1/1980-11/30/01 and NIH/NIBIB R01 EB001973 JSim: A Simulation Analysis Platform, 3/1/02-2/28/07.

