This is a reproduction of the 4 state model of a cardiac sarcomere originally developed by Landesberg and Sideman. The particular version presented here was used in Yaniv, Sivan and Landesberg Am J Physiol 288:H389-H399, 2005.
Description
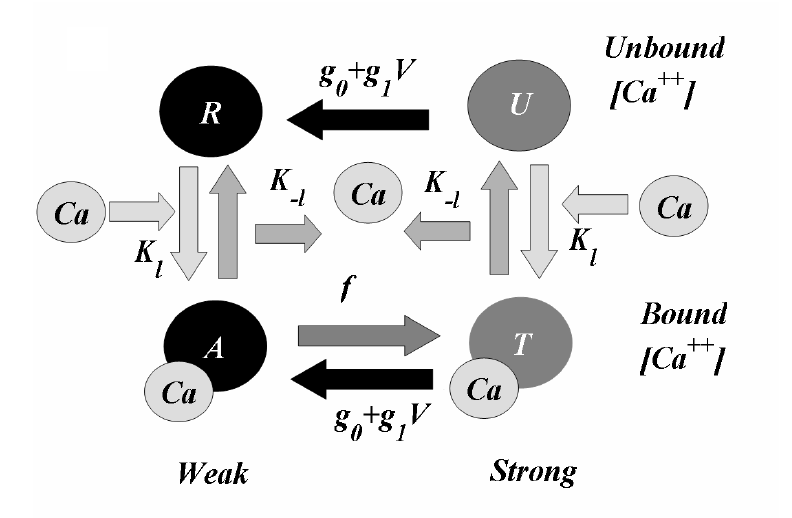
This code represents the simplified Four State model of the sarcomere as developed by Landsberg et al and used by Yaniv in the paper analyzing the sarcomeric control of contraction (AJP 288:H389-H399, 2005, Reference 1 below). The model simplification over previous Landesberg models is that the movement of troponin units between overlap regions is ignored and only the troponin in the single overlap region is considered as the sarcomere extends and contracts. This model describes the regulation of crossbridge cycling and force production present in a single cardiac sarcomere using a four state representation of the troponin regulatory units. Troponin is either in a strong or a weak state and either has calcium bound to it or is unbound. The force generated in the sarcomere is determined by the number of troponin units in the single overlap region that are in the strong bound and unbound states, T and U respectively. This model is a simplification of previous models that considered the states of troponin in the single double and no overlap region of the sarcomere. This model incorporates two feedback mechanisms. The first is a positive feedback mechanism affecting the affinity of troponin for calcium. Calcium bound to troponin increases the chance of further bonding of calcium to other available sites on the troponin regulatory unit and is an example of cooperativity. The second is a negative feedback mechanism and involves the influence of the filament sliding velocity on crossbridge cycling. The parameters given in Yaniv et al. are not sufficient to yield the results presented. Recent communication with the authors and a more complete set of model parameters present in a paper in press allow us to estimate the values for some of the parameters used here. See the model notes for more details.
Equations
The set of ODEs governing the transition of the troponin regulatory unit through its four states is given by:
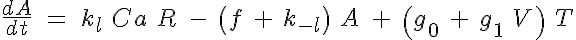
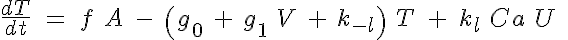
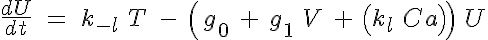
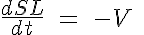
and
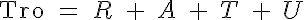
where R, A, T and U are the unbound weak, bound weak, bound strong and unbound strong states of troponin, respectively. Tro is the total concentration of troponin regulatory units, SL is the sarcomere length and V is the sarcomere contraction velocity defined as positive in the case of sarcomere shortening. The negative mechanical feedback is evident in the alteration of the reaction rate from strong to weak states in the form of g0 + g1*V term. The cooperativity mechanism affects the reaction rate from bound to unbound states, k-l, and is given by this set of equations:
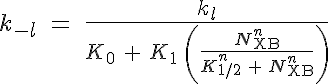
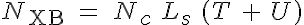
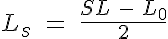
where K0 is the calcium affinity to troponin at rest, K0 + K1 is the maximum caldium affinity, K1/2 is related to the number of force generating crossbridges at 50% of the calcium affinity range, n is the cooperativity coefficient, NXB is the number of force generating crossbridges in the sarcomere, Nc is the number of crossbridges per unit length, Ls is the single overlap length and L0 is a length constant based on the actin and myosin filament lengths.
The sarcomeric force is calculated from:
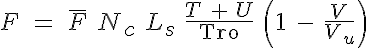
where 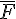 is the average force per crossbridge and Vu is the maximal shortening velocity.
is the average force per crossbridge and Vu is the maximal shortening velocity.
- Download JSim model MML code (text):
- Download translated SBML version of model (if available):
We welcome comments and feedback for this model. Please use the button below to send comments:
Yaniv Y, Sivan R and Landesberg A.;"Analysis of hystereses in force length and
force calcium relations", Am J Physiol Heart Circ Physiol 288:H389-H399, 2005
Yaniv Y, Sivan R and Landesberg A.; "Stability, controllability and observability
of the 'Four State' Model for the sarcomeric control of contraction"
Ann Biomed Eng 34:778-789, 2006
Landesberg A and Sideman S.;"Mechanical regulation of cardiac muscle by coupling
calcium kinetics with cross-bridge cycling: A dynamic model", Am J Physiol Heart
Circ Physiol 267:H779-H795, 1994.
Please cite https://www.imagwiki.nibib.nih.gov/physiome in any publication for which this software is used and send one reprint to the address given below:
The National Simulation Resource, Director J. B. Bassingthwaighte, Department of Bioengineering, University of Washington, Seattle WA 98195-5061.
Model development and archiving support at https://www.imagwiki.nibib.nih.gov/physiome provided by the following grants: NIH U01HL122199 Analyzing the Cardiac Power Grid, 09/15/2015 - 05/31/2020, NIH/NIBIB BE08407 Software Integration, JSim and SBW 6/1/09-5/31/13; NIH/NHLBI T15 HL88516-01 Modeling for Heart, Lung and Blood: From Cell to Organ, 4/1/07-3/31/11; NSF BES-0506477 Adaptive Multi-Scale Model Simulation, 8/15/05-7/31/08; NIH/NHLBI R01 HL073598 Core 3: 3D Imaging and Computer Modeling of the Respiratory Tract, 9/1/04-8/31/09; as well as prior support from NIH/NCRR P41 RR01243 Simulation Resource in Circulatory Mass Transport and Exchange, 12/1/1980-11/30/01 and NIH/NIBIB R01 EB001973 JSim: A Simulation Analysis Platform, 3/1/02-2/28/07.

