React.Seq.FullEnz.Kinetics is the fourth and last of a series to illustrate the influences of capacitances on the duration of transients in the reaction series A --> B --> C --> D--> E in a compartment. It includes the on- and off kinetics for enzyme-substrate binding and release for each reaction; the capacitive delay is the dwell time in the ES complex.
Description
A reaction sequence A-->B-->C-->D-->E can be represented many ways to approximate the biological
form of the reactions. This version takes into account the on-off rates of the enzymatic reactions
resulting in a capacitance within the system which was approximated in the previous model ,MM with
lag (model #425). The model generates progress curves as in a bioreactor, but with additional factor
of a flow through the mixing chamber. The flow term is first order for all solutes in the sequence.
The inflowing initial solute A, the flow, ml/sec, Flow*CinA, equals the sum of the outflow
clearance plus the last reaction E--> ?, i.e. G*E, then the outflow concentration of A in the steady
state goes to 50% of CinA (Do this by setting Flow1 = Flow2 = 0.05 ml/sec and setting GA1 and GA2 also
to 0.05 and run the program. The other reaction rates, to form C, D, and E are set identidal to that
to form B from A, the result is that steady state concentrations for A, B, etc go to 0.5, 0.25, 0.125,
0.0625, and 0.03125, all at one half of its predecessor. This program illustrates the transient
delays between steady state, and the form of the transients. With MM kinetics there is really no
enzyme there, and no binding of the reactants in the process of the reaction. All of the delay is due
to the combination of flow and the reaction, exactly similar to the first order reaction kinetic
model (Model #432). The initial conditions are zero for all solutes so the time constant for the
initial entry would be simply the volume divided by flow, Vol/Flow1, in seconds, if there were no
reaction. The reaction, augmenting the disappearance of A, shortens the time constant so that it
is Vol/(GA + Flow1).
The second transient is due to a step increase (or decrease) in flow at time TFjump (at t=30 s
with this parameters set ('full181028'). The third transient is a step change in the reaction rates at
time TGjump. The Verification Process is the same as for the linear kinetics, and verifies the code
at steady-state with a input solute concentration (CinA) of 1 mM.
The solution to the differential equation for solute A is accomplished using the numerical solvers,
but the steady state solution is also solvable analytically. The three transients, at t = 0, t= 30 s
and t=60 s, are expressed in three analytical equations. Their sum fits exactly the numerical solutions
to the systems equations at steady-state (Anal3). VERIFICATION! Try this out using the difference ODE
solvers: it turns out the DOPRIS5 (an advanced RungeKutta algorithm) gives a more precise fit to the
analytical solution than either of the more powerful stiff solvers, CVODE or Radau. At the steep parts
of the transients DOPRIS5 is still good to 7 decimal digits, while the other supposedly superior solvers,
CVODE and Radau are good only to 5 or 6 decimal digits. In the steady state they all give the same
correct answers.
For solutes B through E, the analytical solutions are more complex, and have not been developed.
For these it is much faster to use the numerical solutions to the ODEs; the analytical solutions for
solute E would be, because of their complexity, much slower to compute, and maybe not even as accurate
as the numerical solutions, even for this rather simple model system. What we know is that the steady
state solution match exactly to the predicted steady state values for A through E.
The intent for this series of models is to account for substrate capacitance in enzymatic networks:
the first model is Linear.Reaction.Sequence (Model 423), without capacitance; the second (Model 424)
is MM.React.Seq, a Michaelis-Menten system without any capacitance. The third, MM.Lag.ReactSeq
(Model 425), adds time lags to account for capacitance, which equals "bound substrate mass / flux of
substrate. The fourth, this model React.Seq.FullEnz.Kinetics (Model 426), abandons the MM
assumptions, and adds the on- and off kinetics for a single uncomplicated reaction.
The Michaelis-Menten Model (#425) that approximates the effect of solute binding to the enzymes
attempts to correct the kinetics of M-M expressions by incorporating lags to represent the buffering
capacitance and consequent delay in transient responses in enzyme systems.
In this model React.Seq.FullEnzKinetics.proj (#426) the capacitance is accounted for by the kinetic
expressions automatically, without using any lag. The approximation in this model is that the enzymes Zs
and their complexes, the ZsS, do NOT get washed out by the flow, but are completely retained in the
compartment. (This is physiologically reasonable only if enzymes are fixed on the wall and diffusion
distances are small.)
The step responses, PLOT 1.StepsA_E, using the unit step input function with CinA = 1 mM (or other level),
are set up for a step increase in flow at t = TFjump and then later at t= TGjump a step in reaction rates.
These are all, necessarily and unavoidably, delayed by the capacitance for substrate in the enzyme-substrate
complex ZsS (where Zs is the enzyme for reacting substrate S). These can be compared with the solutions of
model #425 where the lag is approximated by a concentration-dependent first order time lag. On the PLOT StepA_E
the curve Anal3 (thick black dashes) represents the solutions to the first order kinetic model (#423) and the
unadorned MM model (#424) both of which lack capacitance. The difference between the black dashed curve and
the red curve is due to the capacitance of the enzyme-substrate complex, ZaA.
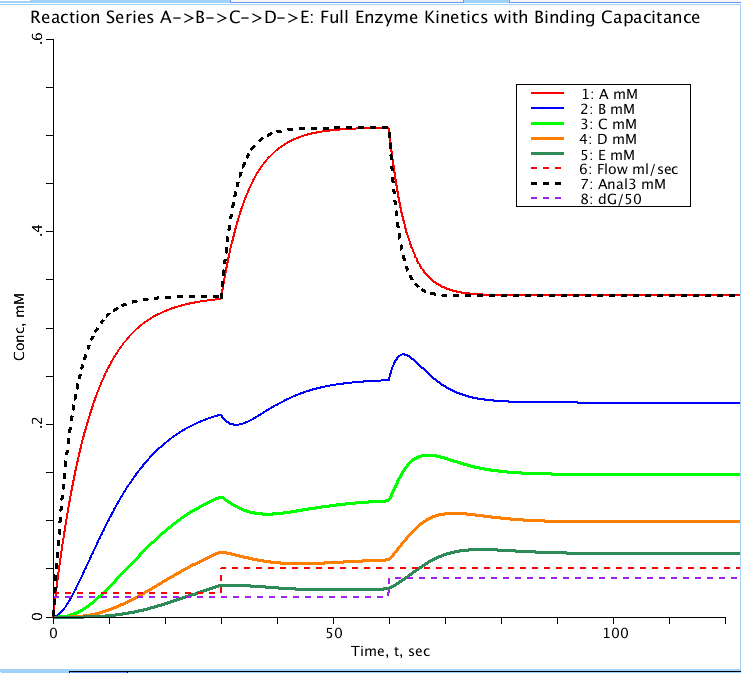
Figure: Progress curves for a sequence of reactions from substrate A to E in a compartment with flow through the compartment. All substrates have initial concentration of zero with A in (CinA) set to 1 mM. Flow doubles from 0.025 to 0.05 ml/sec at t= 30 sec. Substrate consumption factor (dG dimensionless) doubles from 0.05 to 0.1 at t= 60 sec to counter the change in flow. Anal3 is the analytical solution for substrate concentration A(t) for the equivalent first order kinetic model lacking enzyme binding. The delay between Anal3 (black dashes) and A(red curve) is due to the ES capacitance.
Equations
The equations for this model may be viewed by running the JSim model applet and clicking on the Source tab at the bottom left of JSim's Run Time graphical user interface. The equations are written in JSim's Mathematical Modeling Language (MML). See the Introduction to MML and the MML Reference Manual. Additional documentation for MML can be found by using the search option at the Physiome home page.
- Download JSim model MML code (text):
- Download translated SBML version of model (if available):
- No SBML translation currently available.
- Information on SBML conversion in JSim
We welcome comments and feedback for this model. Please use the button below to send comments:
Easterby, JS. A generalized theory of the transition time for sequential enzyme reactions.
Biochem J. 199: 155-161, 1981.
Cascante M, Melendez-Hevia E, Kholodenko B, Sicilia J, and Kacser H. Control analyis
of transit time for free and enzyme-bound metabolites: physiological and
evolutionary significance of metabolic response times. Biochem J 308: 895-899, 1995.
Bassingthwaighte JB. Capacitance in metabolic netowrks. 2019 (in prep for submission to Biophys J)
Please cite https://www.imagwiki.nibib.nih.gov/physiome in any publication for which this software is used and send one reprint to the address given below:
The National Simulation Resource, Director J. B. Bassingthwaighte, Department of Bioengineering, University of Washington, Seattle WA 98195-5061.
Model development and archiving support at https://www.imagwiki.nibib.nih.gov/physiome provided by the following grants: NIH U01HL122199 Analyzing the Cardiac Power Grid, 09/15/2015 - 05/31/2020, NIH/NIBIB BE08407 Software Integration, JSim and SBW 6/1/09-5/31/13; NIH/NHLBI T15 HL88516-01 Modeling for Heart, Lung and Blood: From Cell to Organ, 4/1/07-3/31/11; NSF BES-0506477 Adaptive Multi-Scale Model Simulation, 8/15/05-7/31/08; NIH/NHLBI R01 HL073598 Core 3: 3D Imaging and Computer Modeling of the Respiratory Tract, 9/1/04-8/31/09; as well as prior support from NIH/NCRR P41 RR01243 Simulation Resource in Circulatory Mass Transport and Exchange, 12/1/1980-11/30/01 and NIH/NIBIB R01 EB001973 JSim: A Simulation Analysis Platform, 3/1/02-2/28/07.

